Microwave-assisted extraction of triterpenoid from Abelmoschus sagittifolius (Kurz) Merr roots using deep eutectic solvents
Main Article Content
Abstract
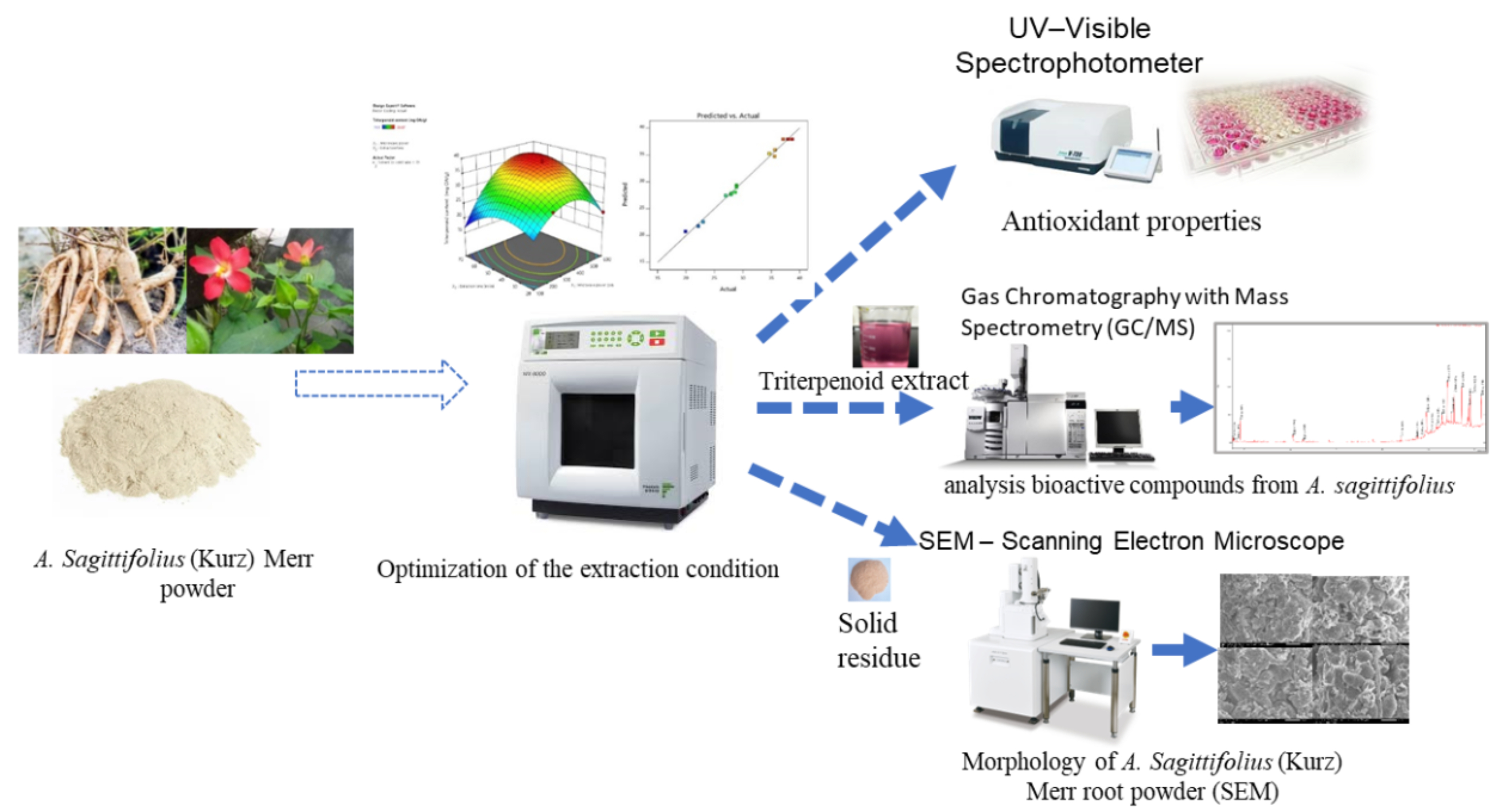
A green and efficient method was developed for extracting triterpenoids from Abelmoschus sagittifolius roots using microwave-assisted extraction (MAE) combined with a choline chloride–citric acid-based deep eutectic solvent (DES). Optimization by response surface methodology identified optimal conditions (365 W, 46 min, 40 mL/g), yielding 39.8 mg/g of triterpenoids, in close agreement with the predicted value (39.5 mg/g). This yield was significantly higher than those obtained by ethanol-based MAE (32.69 mg/g) and Soxhlet extraction (28.14 mg/g). SEM analysis revealed marked cell wall disruption in DES–MAE-treated samples. The extract exhibited strong antioxidant activity (84.56% DPPH and 96.67% ABTS at 0.55 mg/mL) and notable tyrosinase inhibition (IC₅₀ ≈ 0.17 mg/mL), outperforming conventional extracts. GC–MS analysis identified 18 bioactive compounds, predominantly triterpenoids. Overall, DES–MAE represents a sustainable approach for producing triterpenoid-rich extracts for food, cosmetic, and pharmaceutical applications.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
Ngoc TD, Thi Ha MV, Le TN, Thi HV, Anh Nguyen TV, Mechler A, et al. A Potent Antioxidant Sesquiterpene, Abelsaginol, from Abelmoschus sagittifolius: Experimental and Theoretical Insights. ACS Omega. 2022;7:24004–11.
Nguyen PL, Ho Y-L, Le VM, Heinrich M, Chang Y-S. The Vietnamese medicinal and food plant Abelmoschus sagittifolius (Kurz.) Merr., an underestimated resource. Ind Crops Prod. 2024;216(118690):118690.
Ngo MH, Nguyen H, Nguyen TB, Le TD, Le QH. Experimental Study of the Effect of Ultrasound on the Freezing Process of Bo Chinh Ginseng. Appl Sci. 2023;13(1):408.
Chen D-L, Li G, Liu Y-Y, Ma G-X, Zheng W, Sun X-B, et al. A new cadinane sesquiterpenoid glucoside with cytotoxicity from Abelmoschus sagittifolius. Nat Prod Res. 2019;33(12):1699–1704.
Vo TP, Pham TV, Tran TNH, Vo LTV, Vu TT, Pham ND, et al. Ultrasonic-Assisted and Microwave-Assisted Extraction of Phenolics and Terpenoids from Abelmoschus sagittifolius (Kurz) Merr Roots Using Natural Deep Eutectic Solvents. ACS Omega. 2023;8(32):29704–29716.
Hashemifesharaki R, Xanthakis E, Altintas Z, Guo Y, Gharibzahedi SMT. Microwave-assisted extraction of polysaccharides from the marshmallow roots: Optimization, purification, structure, and bioactivity. Carbohydr Polym. 2020;240(116301):116301.
Cai C, Ma J, Han C, Jin Y, Zhao G, He X. Extraction and antioxidant activity of total triterpenoids in the mycelium of a medicinal fungus, Sanghuangporus sanghuang. Sci Rep. 2019;9(1):7418.
Pandey DK, Kaur P. Optimization of extraction parameters of pentacyclic triterpenoids from Swertia chirata stem using response surface methodology. 3 Biotech. 2018;8(3):152.
Chen Y, Xie M-Y, Gong X-F. Microwave-assisted extraction used for the isolation of total triterpenoid saponins from Ganoderma atrum. J Food Eng. 2007;81(1):162–170.
Skarpalezos D, Detsi A. Deep eutectic solvents as extraction media for valuable flavonoids from natural sources. Appl Sci (Basel). 2019;9(19):4169.
Petrochenko AA, Orlova A, Frolova N, Serebryakov EB, Soboleva A, Flisyuk EV, et al. Natural Deep Eutectic Solvents for the Extraction of Triterpene Saponins from Aralia elata var. mandshurica (Rupr. & Maxim.) J. Wen. Molecules. 2023;28(8):3614.
Hikmawanti NPE, Ramadon D, Jantan I, Mun’im A. Natural deep eutectic solvents (NADES): Phytochemical extraction performance enhancer for pharmaceutical and nutraceutical product development. Plants. 2021;10(10):2091.
Oludemi T, Barros L, Prieto MA, Heleno SA, Barreiro MF, Ferreira ICFR. Extraction of triterpenoids and phenolic compounds from Ganoderma lucidum: optimization study using the response surface methodology. Food Funct. 2018;9:209–26.
Bakar MFA, Ismail NA, Isha A, Ling ALM. Phytochemical Composition and Biological Activities of Selected Wild Berries (Rubus moluccanus L., R. fraxinifolius Poir., and R. alpestris Blume). Evid Based Complement Alternat Med. 2016.
Deng Y, Wang X, Zhang C, Xie P, Huang L. Enhanced and Green Extraction of Saponins from Gleditsia sinensis Lam. Pods by Ultrasound-Assisted Deep Eutectic Solvents: Optimization and Comprehensive Characterization. Food Bioproc Tech. 2024.
Vo TP, Ho TAT, Ha NMH, Nguyen DQ. Optimization of ultrasonic and microwave-assisted extraction with natural deep eutectic solvents for enhanced recovery of phenolics and terpenoids from celery leaves. Biomass Conv Bioref. 2025;15:15053–15067.
Vo TP, Ho TAT, Truong KV, Ha NMH, Nguyen DQ. Combining novel extraction techniques with natural deep eutectic solvent to acquire phenolic and terpenoid compounds from Paris polyphylla roots. J Agric Food Res. 2024;18:101544.
Hai TC. Extraction of Chitin from Giant Tiger Prawn (Penaeus monodon) Shrimp Shell Using Deep Eutectic Solvents and Citric Acid. Appl Sci Eng Prog. 2023.
Nekkaa A, Benaissa A, Lalaouna AED, Mutelet F, Canabady-Rochelle L. Optimization of ultrasound-assisted deep eutectic solvent extraction of polyphenols from Rhamnus alaternus for their antioxidant activity. Biomass Conv Bioref. 2025;15:3599–615.
Ali MA, Kaium MA, Uddin SN, Uddin MJ, Olawuyi O, Campbell ADJr, et al. Elucidating the Structure, Dynamics, and Interaction of a Choline Chloride and Citric Acid Based Eutectic System by Spectroscopic and Molecular Modeling Investigations. ACS Omega. 2023;8:38243–38251.
Wei L, Zhang W, Yin L, Yan F, Xu Y, Chen F. Extraction optimization of total triterpenoids from Jatropha curcas leaves using response surface methodology and evaluations of their antimicrobial and antioxidant capacities. Electron J Biotechnol. 2015;18:88–95.
Jemmali Z, Chartier A, Dufresne C, Elfakir C. Optimization of the derivatization protocol of pentacyclic triterpenes prior to their gas chromatography–mass spectrometry analysis in plant extracts. Talanta. 2016;147:35–43.
Vilková M, Płotka-Wasylka J, Andruch V. The role of water in deep eutectic solvent-base extraction. J Mol Liq. 2020;304:112747.
Hao J, Si Q, Wang Z, Jia Y, Fu Z, Zhao M, et al. Optimization of extraction process and dynamic changes in triterpenoids of Lactuca indica from different medicinal parts and growth periods. Molecules. 2023;28(8):3345.
Thong-on W, Pathomwichaiwat T, Boonsith S, Koo-amornpattana W, Prathanturarug S. Green extraction optimization of triterpenoid glycoside-enriched extract from Centella asiatica (L.) Urban using response surface methodology (RSM). Sci Rep. 2021;11(1)22026.
Hai TC, Van Man P, Hong Anh LT. Optimization of ultrasound-assisted extraction of astaxanthin from black tiger shrimp (Penaeus monodon) shells using deep eutectic solvent and ethanol as a co-solvent. Lebenson Wiss Technol. 2024;212:116965.
Desmiaty Y, Hanafi M, Saputri FC, Elya B, Rifai EA, Syahdi RR. Two triterpenoids from Rubus fraxinifolius leaves and their tyrosinase and elastase inhibitory activities. Sci Rep. 2021;11.
Jang WY, Hwang JY, Cho JY. Ginsenosides from Panax ginseng as Key Modulators of NF-κB Signaling Are Powerful Anti-Inflammatory and Anticancer Agents. Int J Mol Sci. 2023;24(7):6119.
Cao TQ, Han JH, Lee H-S, Ha MT, Woo MH, Min BS. Anti-inflammatory and Immunosuppressive Effects of Panax notoginseng. Nat Prod Sci. 2019;25(4):317.
Liang Z. Bioactive components in Panax notoginseng and Panax quinquefolium leaves and their antioxidant, antihypertensive and anti-inflammatory capacities. Ind Crops Prod. 2024;210:118079.